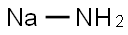Sodium amide
Sodium amino properties
| Fusing point | 210 °C (lit.) |
|---|---|
| Boiling point | 400 °C (lit.) |
| density | 1.39 g/cm3 (25℃) |
| Flash point | 85 °F |
| Storage condition | Store below +30°C. |
| solubility | React with H2O |
| form | crystallization |
| colour | Off-white powder |
| Odor | Ammonia odor |
| Water solubility | REACTS VERY VIOLENTLY, EVEN EXPLOSIVELY |
| sensibility | Air & Moisture Sensitive |
| Merck | 14,8576 |
| stability | Flammable. Reacts violently with water to produce highly toxic smoke. In case of a fire, don't use water, but use baking soda to put it out. If heated or in contact with air or oxygen for a long time, explosive peroxides may form. Incompatible with water and aqueous solutions, carbon dioxide, halogenated hydrocarbons |
| CAS database | 7782-92-5(CAS DataBase Reference) |
| EPA Chemical information | Sodium amide (Na(NH2)) (7782-92-5) |
Post-treatment is if the amount is not large, it can be directly quenched with water, and if the amount is large, slowly add water. It is suspended in dry tetrahydrofuran, stirring and slowly adding ethanol or isopropyl alcohol until no more hydrogen is released and clarified.
Sodium amino, also known as sodium amino or sodium ammonia, is a white or olive green crystalline powder, with ammonia flavor, chemical formula NaNH2, molecular weight 39.01, melting point of 210℃, boiling point of 400℃. When heated to 500 ~ 600℃, it is decomposed into sodium, nitrogen and hydrogen. It reacts violently with water to form sodium hydroxide and gives off ammonia gas. It is slightly soluble in liquid ammonia and reacts slowly with alcohol. Quickly absorb carbon dioxide and water in the air, should be sealed storage. Liquid can dissolve metal magnesium, zinc, molybdenum, tungsten and quartz, glass, silicate and other substances. An ionic compound consisting of a sodium ion and an ammonia molecule that has removed a hydrogen atom to form the -1 valence group aminoNH-2. Sodium aminide is flammable, explosive, corrosive and deliquable. Because amino group has a lone electron pair, easy to bind protons, so in water strongly hydrolyzed, its aqueous solution is alkaline: NaNH2+H2O=NaOH+NH3. Soluble in liquid ammonia and hot ethanol. Dust is toxic. Severe skin, eye and respiratory irritation. Can cause corrosive burns on contact with skin. Used as an ammoniating agent in organic synthesis; Condensation accelerator; Dehydrating agent, dehalogenating agent; Initiators for polymerization and raw materials for the manufacture of hydrazine, sodium cyanide, azide, cyanide, hydrazine and indigo.Sodium amino is easily oxidized in the air, generating a layer of yellow oxidation products on the surface. Some of these oxidation products are explosive and may detonate due to friction or heat. When sodium aminoide is heated in a vacuum to 300 ~ 330 ° C, it is decomposed into nitrogen, sodium, hydrogen and ammonia. Therefore, it should be sealed in a bottle filled with an inert atmosphere to avoid contact with air, water or fire to avoid explosion and fire. Sodium amino and carbon monoxide reaction can produce sodium cyanide, can facilitate this reaction to produce sodium cyanide. Sodium amino is usually used as a condensation accelerator, dehydrating agent, alkylating agent in organic synthesis. In the industry, sodium amination is obtained by the reaction of molten sodium and gaseous ammonia at 300 ~ 400℃, and in the laboratory, it is obtained by the reaction of sodium and liquid ammonia in the presence of a catalyst at room temperature. The dust is toxic and can cause caustic burns and can severely irritate the eyes, skin and respiratory system.
When disposing of escaped materials, wear a gas mask and gloves, mix with dry sand and soil and send it to an open place, react with a large amount of water, and then release it into the wastewater system. The polluted ground should also be washed with water; Skin contact, eye irritation, should be immediately washed with water; Stray into the mouth, should immediately gargle, drinking water and vinegar or 1% acetic acid, and then sent to the hospital for treatment.
2Na+2NH3→2NaNH2+H2↑
Safety information
| Dangerous goods mark | F,C,Xi,N |
|---|---|
| Hazard class code | 14/15-19-34-20/21-10-67-65-63-48/20-11-36/37-15/29-14-50-29 |
| Safety statement | 26-43-45-62-7/8-43D-36/37/39-27-16-61 |
| Dangerous goods transport number | UN 3129 4.3/PG 2 |
| WGK Germany | 2 |
| F | The 3-10-23 |
| Spontaneous combustion temperature | 450 °C |
| TSCA | Yes |
| Customs code | 2853 90 90 |
| Danger class | 4.3 |
| Packaging category | II |
| Toxic substance data | 7782-92-5(Hazardous Substances Data) |
| provider | Language |
|---|---|
|
Chinese
|
|
|
English
|
|
|
English
|
|
|
Chinese
|
|
|
English
|