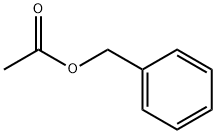
Benzyl acetate
Benzyl acetate properties
| Fusing point | -51 °C (lit.) |
|---|---|
| Boiling point | 206 °C (lit.) |
| density | 1.054 g/mL at 25 °C (lit.) |
| Vapor density | 5.1 |
| Vapor pressure | 23 mm Hg ( 110 °C) |
| Refractive index | n
|
| FEMA | 2135 | BENZYL ACETATE |
| Flash point | 216 °F |
| Storage condition | -20°C |
| solubility | ≥35.7 mg/mL in ethanol; DMSO ≥49.4 mg/mL |
| form | liquid |
| colour | Colorless liquid |
| Odor | Sweet floral scent |
| explosive limit | 0.9 8.4% (V) |
| Flavor type | floral |
| Water solubility | < 0.1g /100 mL at 23 ºC |
| Merck | 14,1123 |
| JECFA Number | 23 |
| BRN | 1908121 |
| Dielectric constant | 5.1 (21 ° C) |
| Exposure limit | ACGIH: TWA 10 ppm |
| LogP | 1.96 at 25℃ |
| CAS database | 140-11-4(CAS DataBase Reference) |
| (IARC) classification of carcinogens | 3 (Vol. 40, Sup 7, 71) 1999 |
| NIST Chemical information | Benzyl ethanoate(140-11-4) |
| EPA Chemical information | Benzyl acetate (140-11-4) |
Add 0.5 g benzyl chloride and ammonium acetate (0.77 mmol) together to 5 ml glycerin. After mixing, the reaction mixture is heated to 80°C in an oil bath. Let the reaction run for 1 hour. Let the mixture cool. Separate the product. After cooling, the mixture was extracted with 5mL of petroleum ether. Concentrate the organic phase under reduced pressure. The crude oil was analyzed by gas chromatography with HP-I column. The title compound benzyl acetate was obtained by evaporation under reduced pressure.
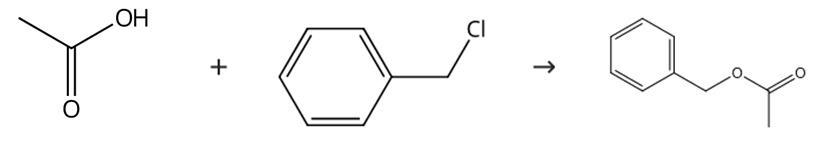
The synthetic route of benzyl acetate is shown
According to method 1 of ester content determination method (OT-18). The sample size was 900mg. Equivalent factor (e) in the calculation or determined by gas chromatography (GT-10-4) with a non-polar column. It is rapidly hydrolyzed and metabolized in animals.Adl 0~5mg/kg(FAO/WHO, 1994).
LD 502490-3696 mg/kg(rat, oral). FEMA(mg/kg) : Soft drinks 7.8; Cold drinks 14; Candy 34; Baked goods 22; Pudding 23; Gum 760.
Moderation is limited (FDA§ 172.515, 2000).
Under the catalysis of sulfuric acid, benzyl alcohol and glacial acetic acid were mixed and heated to esterify. After the reaction, it was first neutralized with alkali and then washed with water until neutral. After drying with anhydrous calcium chloride, the product is distilled under reduced pressure. A small amount of boric acid is added before distillation so that excess benzyl alcohol, which forms a high boiling point benzyl borate, remains in the still.
CH3COOH+C6H5CH2OH[H2SO4]→CH3CHOOCH2C6H5+H2O
The finished product is made by the reaction of chlorobenzyl with sodium acetate. The chlorine in the product must be removed. The yield of this method can reach 80%.
CH3COONa+C6H5CH2ClCH3COOCH2C6H5+NaCl
1kg benzyl alcohol and 2kg anhydrous sodium acetate were added to 1.2kg acetic anhydride successively, and slowly heated to l00℃. After the reaction was moderate, 40g anhydrous sodium acetate was added, and then reflux reaction was carried out for 8h. After cooling, wash with water, neutralize with 10% NaOH solution, then wash until neutral, and finally dry with anhydrous calcium chloride, and then distill the product under vacuum.
(CH3CO)2+C6H5CH2OH[NaAc]→C3H7COOCH2C6H5+CH3COOH
It is formed by esterification of benzyl alcohol with acetic anhydride or acetic acid and purified by distillation.
Safety information
| Dangerous goods mark | Xi |
|---|---|
| Hazard class code | 36/37/38 |
| Safety statement | 26-37/39-24/25 |
| Dangerous goods transport number | 2810 |
| WGK Germany | 1 |
| The RTECS | AF5075000 |
| Spontaneous combustion temperature | 862 °F |
| TSCA | Yes |
| Customs code | 29153950 |
| Toxic substance data | 140-11-4(Hazardous Substances Data) |
| Toxicity | LD50 orally in rats: 2490 mg/kg (Jenner) |
| provider | Language |
|---|---|
|
Chinese
|
|
|
English
|
|
|
English
|
|
|
English
|
|
|
Chinese
|
|
|
English
|
| Update date | Product number | Product name | CAS number | package | Price |
|---|---|---|---|---|---|
| 2024/04/30 | B23472 | Benzyl acetate, 99% Benzyl acetate, 99% |
The 140-11-4 | 500g | 351 yuan |
| 2024/04/30 | B23472 | Benzyl acetate Benzyl acetate |
The 140-11-4 | 2500g | 991 yuan |